WNT-4 real? A proposed paracrine player in β-cell function and glycemic control
by Ulrik Larsen
Diabetes is a multifactorial disorder commonly characterized by the loss or dysfunction of pancreatic β-cell, which play a central role in insulin secretion. β-cells have been shown to be heterogeneous, differing in glucose sensing, insulin secretion, and gene expression. They communicate with other endocrine cells via paracrine signaling and with each other through gap junctions. While gap junctions are well established in β-cell coordination, the role of paracrine signaling remains less understood.
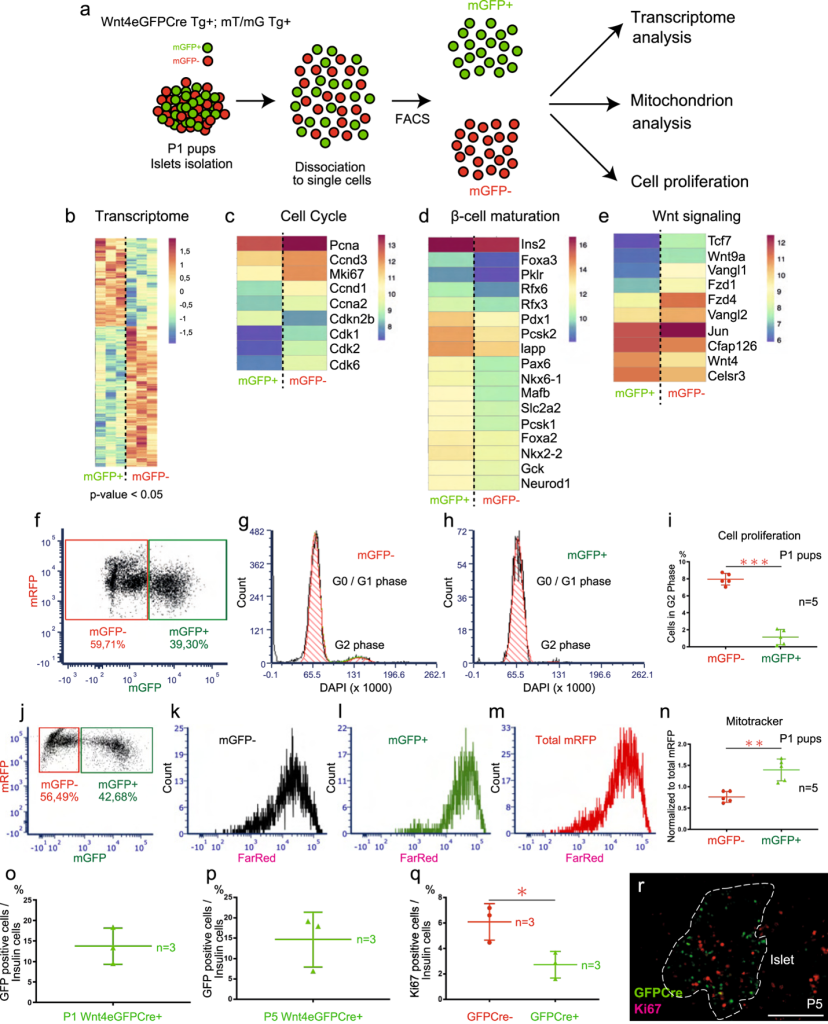
This study explores the role of WNT4 in β-cell function and adaptability using Wnt4-eGFPCre mice for lineage tracing and an inducible PDXcreER model causing WNT4 downregulation in mature mice. Sorting of GFP-positive and negative cells from isolated islets revealed that WNT4-high cells are more mature, whereas WNT4-low cells are more proliferative. Conditional inactivation of Wnt4 in adult mice led to impaired glucose tolerance and elevated glycemia, not due to reduced β-cell number or size but rather through decreased ATP/ADP ratios and calcium signaling. Interestingly, these effects were sex-dependent, with males being more affected.
The study also highlights how β-cell function is influenced by other extrinsic signals such as noradrenaline (neurons), SST (δ-cells), GCG (α-cells), GLP-1 (L- and α-cells), and GIP (K-cells). Here, WNT4 is proposed as a physiological sensor that helps β-cells adapt their function to metabolic demands, contributing to glycemic regulation between themselves. As another study (https://www.nature.com/articles/s42255-024-01114-8) showed that β-cells can self-regulate even in the absence of other islet cell types further investigation into paracrine signaling from β-cells themselves could be a valuable for promoting β-cell function. A final interesting observation is the briefly discussed “compensation” of Wnt5b when Wnt4 is knocked out. This could possibly as a stress response, like in our moderate ablation dataset during proliferation peak, though here it was mostly to discuss why some Wnt4 targets were not significantly downregulated (such as the planar cell polarity pathway). However, in this case, Wnt4 knockout β-cells did not cause increase in proliferation.
Continue your reading here:
Katsumoto K, Yennek S, Chen C, Silva LFD, Traikov S, Sever D, Azad A, Shan J, Vainio S, Ninov N, Speier S, Grapin-Botton A. (2022). Wnt4 is heterogeneously activated in maturing β-cells to control calcium signaling, metabolism and function. Nat Commun. 2022 Oct 21; 13(1):6255. doi: 10.1038/s41467-022-33841-5.